Two gasdermins join forces to fight plague
In a new study published in “PNAS”, Kaiwen Chen and Benjamin Demarco from the lab of Prof. Petr Broz, report that two pore-forming cell death executors – gasdermin-D and gasdermin-E – play distinct roles in macrophages/neutrophils in defense against Yersinia.
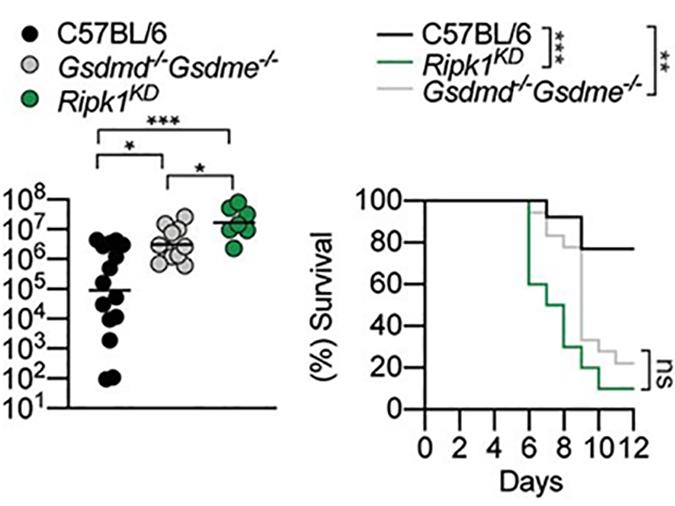
Pathogenic Yersinia are a group of Gram-negative extracellular bacteria that causes disease ranging from gastroenteritis (Yersinia pseudotuberculosis) to plague (Y. pestis). To avoid detection by the host innate immune system, these bacteria inject the effector YopJ, which block proinflammatory cytokine production. To counteract this, detection of YopJ activity by myeloid cells induces the assembly of a cytoplasmic death–inducing complex that comprises RIPK1, FADD and caspase-8. Normally, activation of this platform would induce apoptotic cell death, but during Yersinia infection the platform is re-routed to induce pyroptosis, a pro-inflammatory type of necrosis. Researchers from the Broz lab now show that Rip1-dependent pyroptosis requires two gasdermin family members, GSDMD and GSDME, which are activated by Rip1 indifferent immune cells, macrophages resp. neutrophils. The combined action of both gasdermins is required to drive inflammation and secretion of IL-1b and thus clear Yersinia infections.
https://www.pnas.org/content/118/28/e2101189118
Figure: Ripk1 catalytic dead (KD) and GSDMD/E double-deficient mice display significantly impaired restriction of Yersinia replication and succumb to the infection.