Cell death executor Gasdermin-D has both protective and detrimental functions
In a new study published in “Science Advances”, Benjamin Demarco and Kaiwen Chen from the lab of Prof. Petr Broz, uncover the physiological relevance of a new signaling axis connecting apoptosis signaling to the pyroptosis executor gasdermin-D.
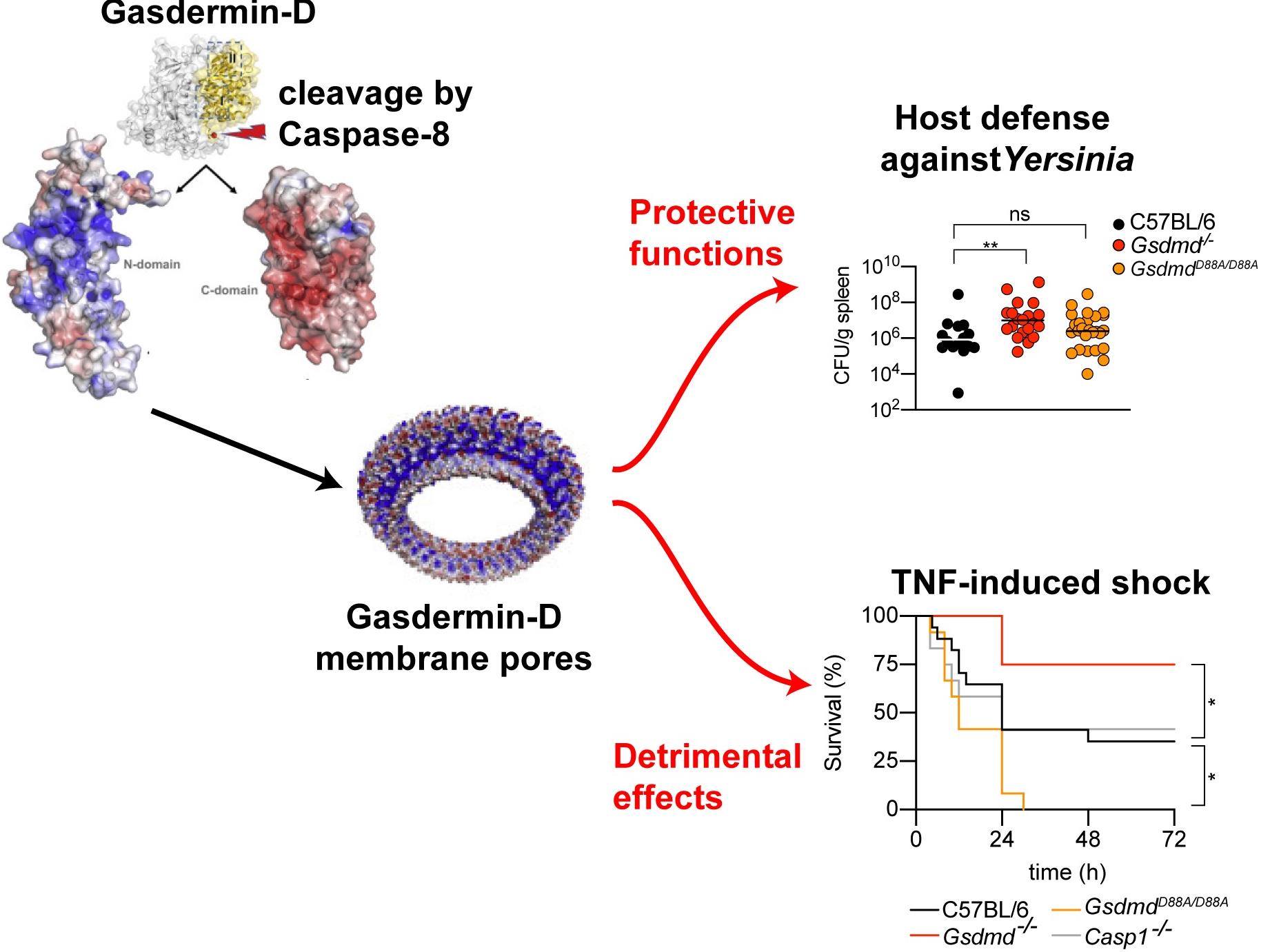
Apoptosis is crucial for embryonic development, removal of autoreactive lymphocytes and clearance of damaged or superfluous cells. Since phagocytic cells rapidly remove apoptotic cells to maintain cellular homeostasis without triggering a proinflammatory response, apoptosis has been assumed to be immunologically silent. In recent years however, mounting evidence suggested that apoptotic macrophages, dendritic cells and neutrophils can nevertheless cause inflammation. The recent discovery of a new signaling axis in which apoptotic caspases cleave and activate gasdermin-D, the executor of a programmed necrotic cell death (pyroptosis), provided a possible explanation for how apoptosis becomes proinflammatory (Chen & Demarco et al. EMBOJ 2019, Orning et al. Science 2018, Sarhan et al. PNAS 2018). Yet if this axis is actually physiologically significant remained unknown.
In the current study, Demarco et al. show that caspase-8-cleaved gasdermin-D is essential to provide protection against Yersinia species, a genus of bacteria that cause a number of diseases ranging from simple enterocolitis (Y. pseudotuberculosis/enterocolitica) to plague (Y. pestis). The authors found that GSDMD-driven pyroptotic cell death promotes anti-Yersinia defense, suggesting that this new discovered CASP8-GSDMD axis plays a crucial role in pathogen clearance.
In contrast to this beneficial function, the authors also report that GSDMD-driven cell death can become the driver of pathological inflammation and even death in a murine TNF-induced shock model. Caspase-8-dependent GSDMD activation and cell lysis was found to be the cause of TNF-induced lethality in vivo, whereas caspase-1 and the proinflammatory cytokines did not contribute to mice susceptibility. In conclusion, the study revealed GSDMD as a potential therapeutic target in inflammatory diseases associated with aberrant TNF signaling such as arthritis, Chron`s disease, and systemic inflammatory response syndrome.