New publication by the van der Meer lab
An analog to digital converter controls bistable transfer competence development of a widespread bacterial integrative and conjugative element
Mobile DNA elements are pieces of genetic material that can jump from one bacterium to another, and even across species. They are often useful to their host, for example carrying genes that allow bacteria to resist antibiotics. In a new publication of the van der Meer group, Nicolas Carraro et al. set out to identify the molecular mechanisms employed by some mobile DNA to control its host.
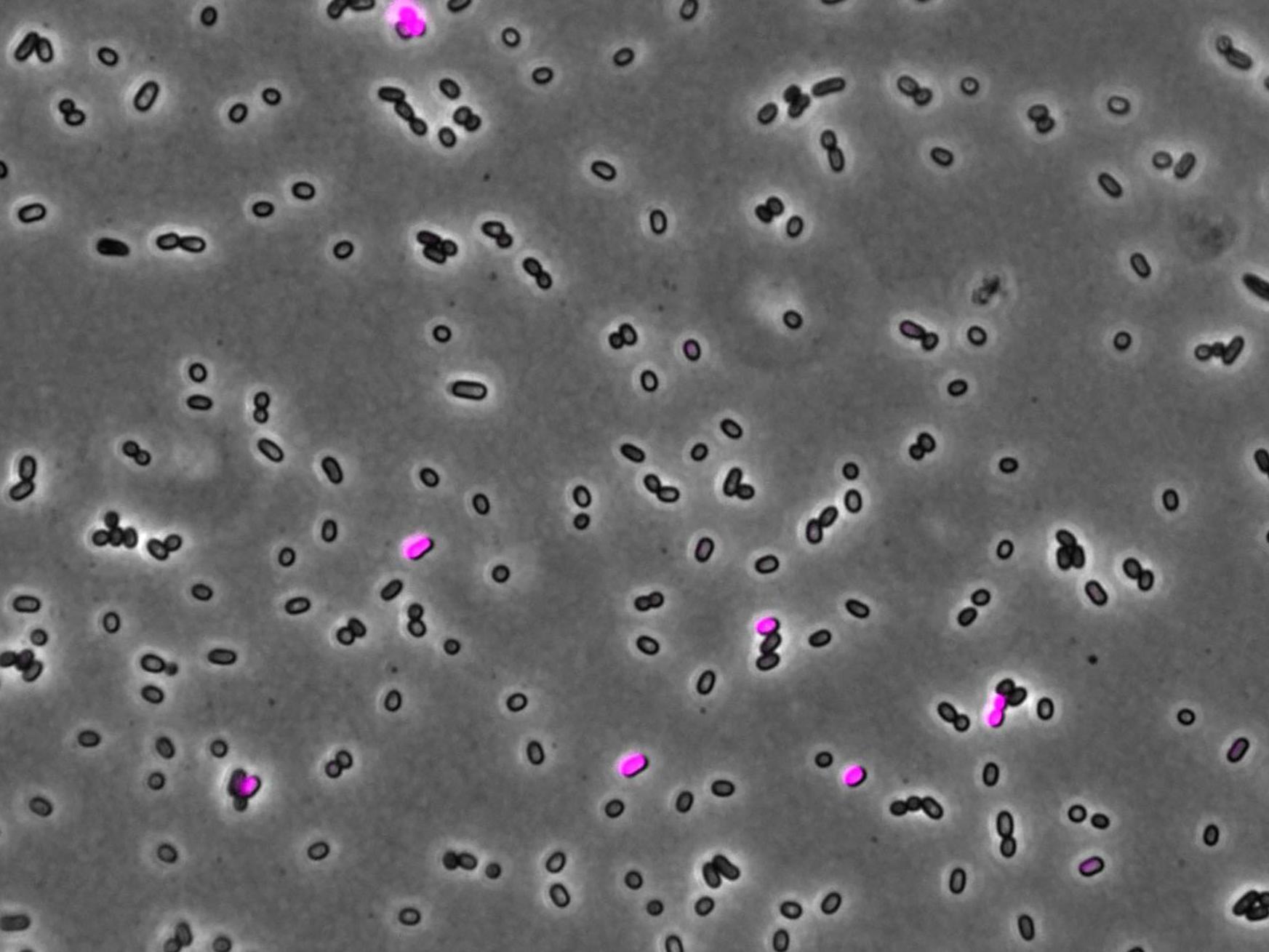
One example of bacterial mobile DNA is the ICEclc element. Usually, ICEclc sits passively within the bacterium’s own DNA, but in a small number of cells, it takes over, hijacking its host to multiply and to get transferred to other bacteria. Cells that can pass on the elements cannot divide, and so this ability is ultimately harmful to individual bacteria. Carrying ICEclc can therefore be positive for a bacterium but passing it on is not in the cell’s best interest. On the other hand, mobile DNAs like ICEclc have evolved to be disseminated as efficiently as possible. To shed more light on this tense relationship,
Experiments using mutant bacteria revealed that for ICEclc to successfully take over the cell, a number of proteins needed to be produced in the correct order. In particular, a protein called BisDC triggers a mechanism to make more of itself, creating a self-reinforcing ‘feedback loop’.
Mathematical simulations of the feedback loop showed that it could result in two potential outcomes for the cell. In most of the ‘virtual cells’, ICEclc ultimately remained passive; however, in a few, ICEclc managed to take over its hosts. In this case, the feedback loop ensured that there was always enough BisDC to maintain ICEclc’s control over the cell. Further analyses suggested that this feedback mechanism is also common in many other mobile DNA elements, including some that help bacteria to resist drugs.
These results are an important contribution to understand how mobile DNAs manipulate their bacterial host in order to propagate and disperse. In the future, this knowledge could help develop new strategies to combat the spread of antibiotic resistance.