Mutations in the palm domain disrupt modulation of acid-sensing ion channel 1a currents by neuropeptides
Bargeton, B., Iwaszkiewicz, J., Bonifacio, G., Roy, S., Zoete, V., and Kellenberger, S. (2019). Scientific Reports 9(1), 2599. doi: 10.1038/s41598-018-37426-5.
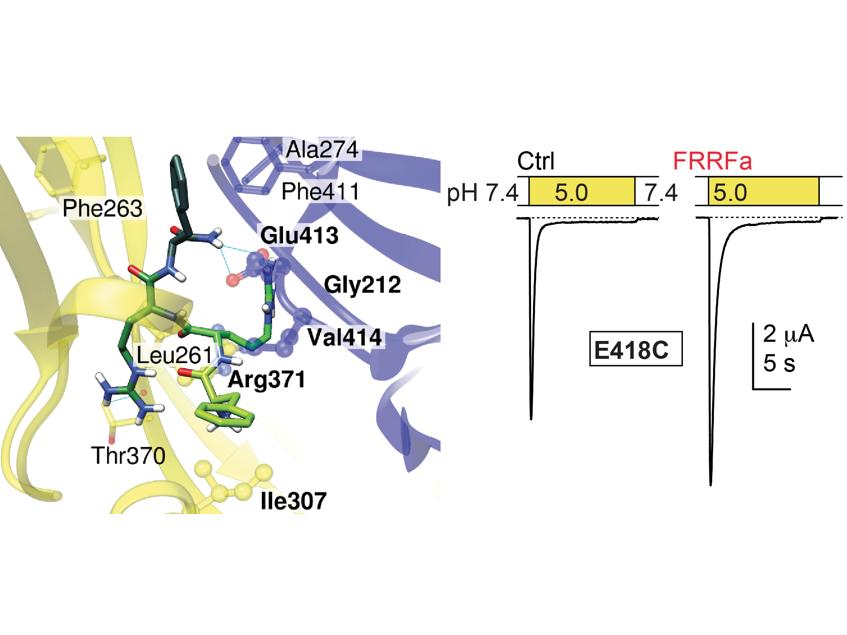
Identification of a modulator binding site on acid-sensing ion channels
A recent study by the team of Stephan Kellenberger identified the binding site of a small modulatory peptide on Acid-sensing ion channels (ASICs). The ASICs are located in the cell membrane of neurons. They can affect the neuronal activity in situations that are accompanied by changes in the acidity of tissues during ischemia and inflammation, or during high neuronal activity. ASICs were shown to contribute to pain sensation, to the sensation of fear, to learning and to neurodegeneration after ischemic stroke. It is thought that ASIC modulators play crucial roles for the activity of ASICs in these situations.
The present study, published on February 22 in “Scientific Reports”, was carried out in collaboration with Justyna Iwaszkiewicz and Vincent Zoete of the Swiss Institute of Bioinformatics. The study was initiated by in silico docking of the peptide to a structural model of the channel, leading to the identification of several possible peptide binding sites. Amino acid residues of the predicted binding pockets were mutated, and it was measured in functional experiments how much such mutations affected the modulation of ASIC currents by the peptide. This approach identified a central part of the channel as the peptide binding site. These results identify a modulatory site on ASICs, and constitute a basis for the investigation of the mode of action of these peptides, and the development of new ASIC modulators.