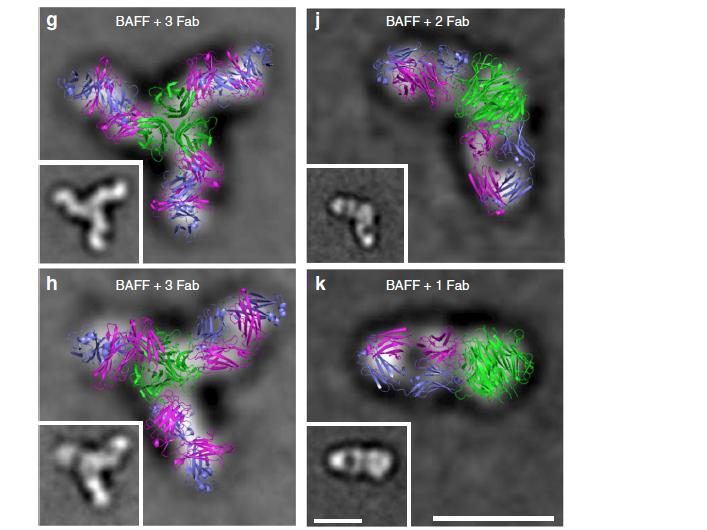
A loop region of BAFF controls B cell survival and regulates recognition by different inhibitors
Michele Vigolo, Melissa G. Chambers, Laure Willen, Dehlia Chevalley, Klaus Maskos, Alfred Lammens, Aubry Tardivel, Dolon Das, Christine Kowalczyk-Quintas, Sonia Schuepbach-Mallepell, Cristian R. Smulski, Mahya Eslami, Antonius Rolink, Edith Hummler, Eileen Samy, Yves Fomekong Nanfack, Fabienne Mackay, Maofu Liao, Henry Hess, Xuliang Jiang & Pascal Schneider; NATURE COMMUNICATIONS | (2018) 9:1199
The B cell survival factor (TNFSF13B/BAFF) is often elevated in autoimmune diseases and is
targeted in the clinic for the treatment of systemic lupus erythematosus. BAFF contains a
loop region designated the flap, which is dispensable for receptor binding. Here we show that
the flap of BAFF has two functions. In addition to facilitating the formation of a highly active
BAFF 60-mer as shown previously, it also converts binding of BAFF to TNFRSF13C (BAFFR)
into a signaling event via oligomerization of individual BAFF-BAFFR complexes. Binding and
activation of BAFFR can therefore be targeted independently to inhibit or activate the function
of BAFF. Moreover, structural analyses suggest that the flap of BAFF 60-mer temporarily
prevents binding of an anti-BAFF antibody (belimumab) but not of a decoy receptor (atacicept).
The observed differences in profiles of BAFF inhibition may confer distinct biological
and clinical efficacies to these therapeutically relevant inhibitors.